Clinical Case Study: Post-Stroke Neural Transmission Disorder
This case study represents a typical long-term post-stroke patient profile frequently encountered. outside the acute hospital setting and inadequately addressed by conventional diagnostic frameworks.
- Patient Profile: Male, middle-aged, history of ischemic stroke approximately 12–24 months prior.
- Motor Impairment: Persistent unilateral weakness, inability to fully open the hand, reduced grip strength, heavy gait with impaired foot clearance, and inability to ambulate independently.
- Involuntary Symptoms: Limb tremor, spasticity, episodic muscle stiffness, and abnormal toe flexion during gait.
- Cranial Nerve Involvement: Mild facial asymmetry and speech articulation difficulty.
Despite stable neuroimaging findings, the patient demonstrated progressive functional deterioration, highlighting the limitations of imaging-centered follow-up alone.
---Physiological and Neurological Data Collected
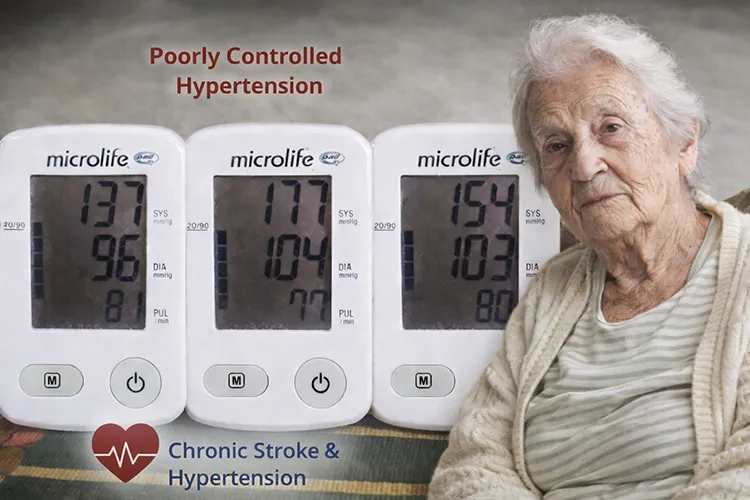
- Multisite Blood Pressure: Significant asymmetry between left and right upper limbs, and between upper and lower extremities.
- Autonomic Indicators: Elevated resting blood pressure variability and inconsistent pulse response.
- Motor Signal Integrity: Delayed voluntary activation and poor motor coordination despite preserved muscle mass.
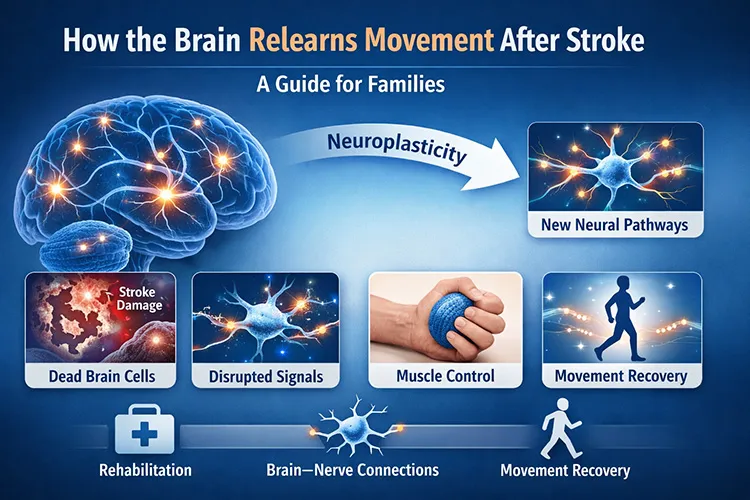
These findings suggest disrupted neural transmission rather than primary muscular pathology.
---Algorithmic Analysis and Output
The diagnostic algorithm processed the collected data through a structured, multi-layered framework.
- Feature Extraction: Detection of asymmetric autonomic control, impaired corticospinal signaling, and maladaptive neuroplastic patterns.
- Neural Pathway Mapping: High likelihood of chronic corticospinal tract disruption with secondary extrapyramidal involvement.
- Severity Scoring: Moderate-to-high functional impairment with elevated risk of secondary neurological events.
The algorithm identified hidden physiological instability not captured by routine clinical follow-up.
---FDA SaMD Description (Software as a Medical Device)
This software qualifies as a Clinical Decision Support Software under the FDA SaMD framework, designed to assist healthcare professionals in neurological assessment and long-term stroke management.
- Intended Use: To analyze physiological and neurological input data in post-stroke patients and provide decision-support insights related to neural pathway integrity, functional impairment severity, and secondary stroke risk.
- Intended User: Licensed healthcare professionals, rehabilitation specialists, and trained clinical staff.
- Input Data: Multisite blood pressure readings, heart rate metrics, motor performance indicators, speech and swallowing observations, and clinician-entered functional assessments.
- Core Algorithm Function: Multidimensional feature extraction, neural pathway correlation, severity scoring, and generation of clinical decision support outputs.
- Output: Risk stratification scores, functional impairment classification, and non-prescriptive rehabilitation and monitoring recommendations.
Risk Management and Regulatory Positioning
- Clinical Role: The software does not provide autonomous diagnosis or treatment decisions.
- Regulatory Alignment: Designed in accordance with FDA guidance on Clinical Decision Support Software and IMDRF SaMD risk categorization principles.
- Data Safety: No direct patient-identifiable data required for algorithmic operation.
Clinical Value Proposition
This SaMD enables early identification of chronic neural transmission disorders in post-stroke patients, bridging the gap between imaging stability and functional decline.
- Home and Community Monitoring: Supports long-term patient follow-up beyond hospital discharge.
- Personalized Rehabilitation Planning: Aligns therapy with neural pathway dysfunction rather than surface-level symptoms.
- Secondary Stroke Prevention: Detects physiological instability associated with recurrent cerebrovascular risk.
Conclusion: This software represents a clinically relevant, regulatory-aligned diagnostic support solution for long-term stroke management and neurorehabilitation.